The ICHOM Set of Patient-Centered Outcome Measures for Overactive Bladder is the result of hard work by a group of leading physicians, measurement experts and patients. It represents the outcomes that matter most to patients who have had an Overactive Bladder. We urge all providers around the world to start measuring these outcomes to better understand how to improve the lives of their patients.
- Tracked via the ICIQ-OAB module
- Tracked via the OAB-q SF
- Tracked via the ICIQ-MLUTSsex or the ICIQ-FLUTSsex
- Tracked via the Treatment Benefit Scale
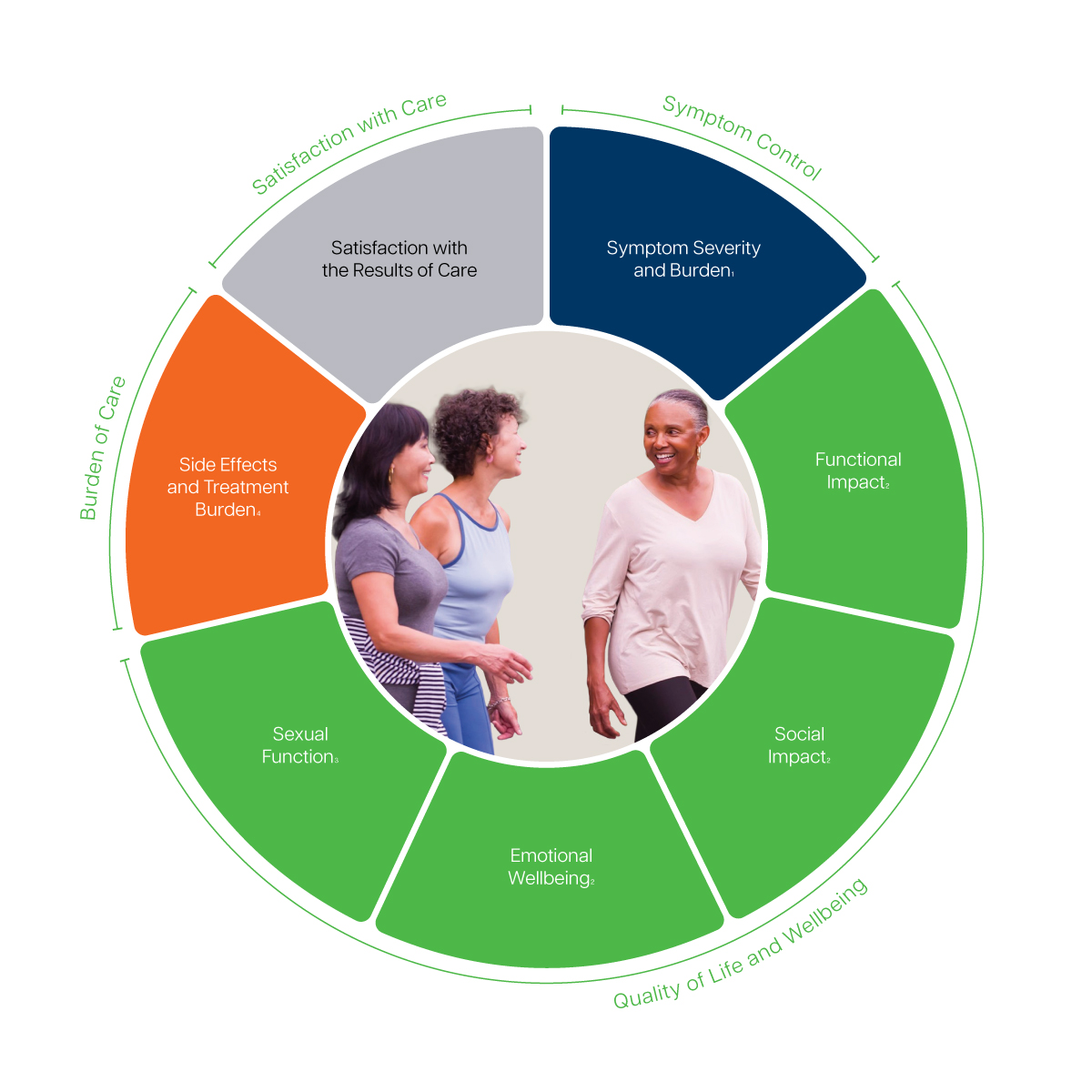

All of the basic information you need to implement ICHOM Sets is available in the links above. However, for a faster, more easily integrated process, we offer digitised versions of the Set measures. These are designed for immediate upload to existing EMR systems. This is available as part of our Accreditation package, which recognises your work in putting patient outcomes at the heart of treatment.
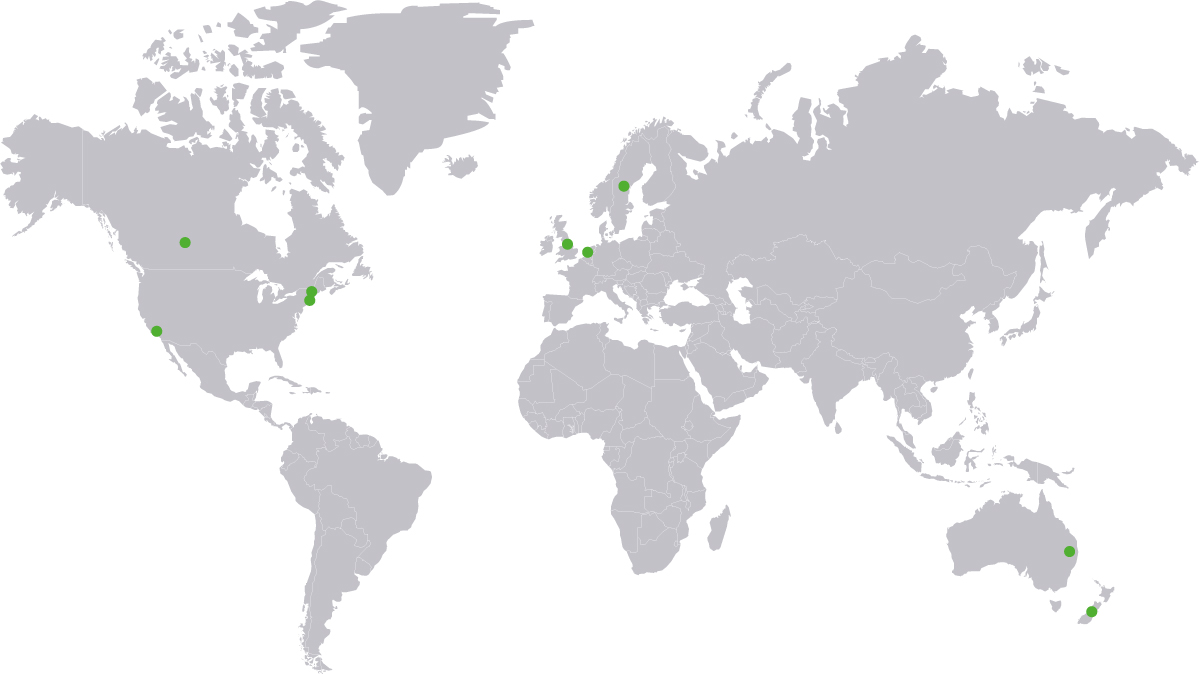
Team that developed this set
AUSTRALIA
George Szonyi | Sydney Local Health District
Kate Moore | St George Hospital, University of NSW
CANADA
Adrian Wagg | University of Alberta
NETHERLANDS
John Heesakkers | UMC St Radboud
NEW ZEALAND
Peter Herbison | Dunedin School of Medicine
SWEDEN
Ian Milsom | Sahlgrenska University Hospital
UNITED KINGDOM
Linda Cardozo | King’s College Hospital NHS
Nikki Cotterill | Bristol Urological Institute
Philip Toozs-Hobson | Birmingham Women’s NHS Foundation Trust
UNITED STATES
Anita Anderson*
Jennifer Anger | Cedars-Sinai Medical Center
Caroline Foust-Wright | Massachusetts General Hospital
Ann Gormley | Hitchcock Dartmouth MC
Jessica McKinney | Marathon PT
Abraham Morse | Boston Urogyn
Samantha Pulliam | Massachusetts General Hospital
*Patient representative
Are you implementing ICHOM Sets?
If your are implementing ICHOM Sets, please help us understand more about your journey by filling in our Implementation Survey. Click on the link below to complete:
Implementation Map
We would like to add you to our Implementation Map if you are implementing or have implemented ICHOM Sets. Please click on the button below for more information.